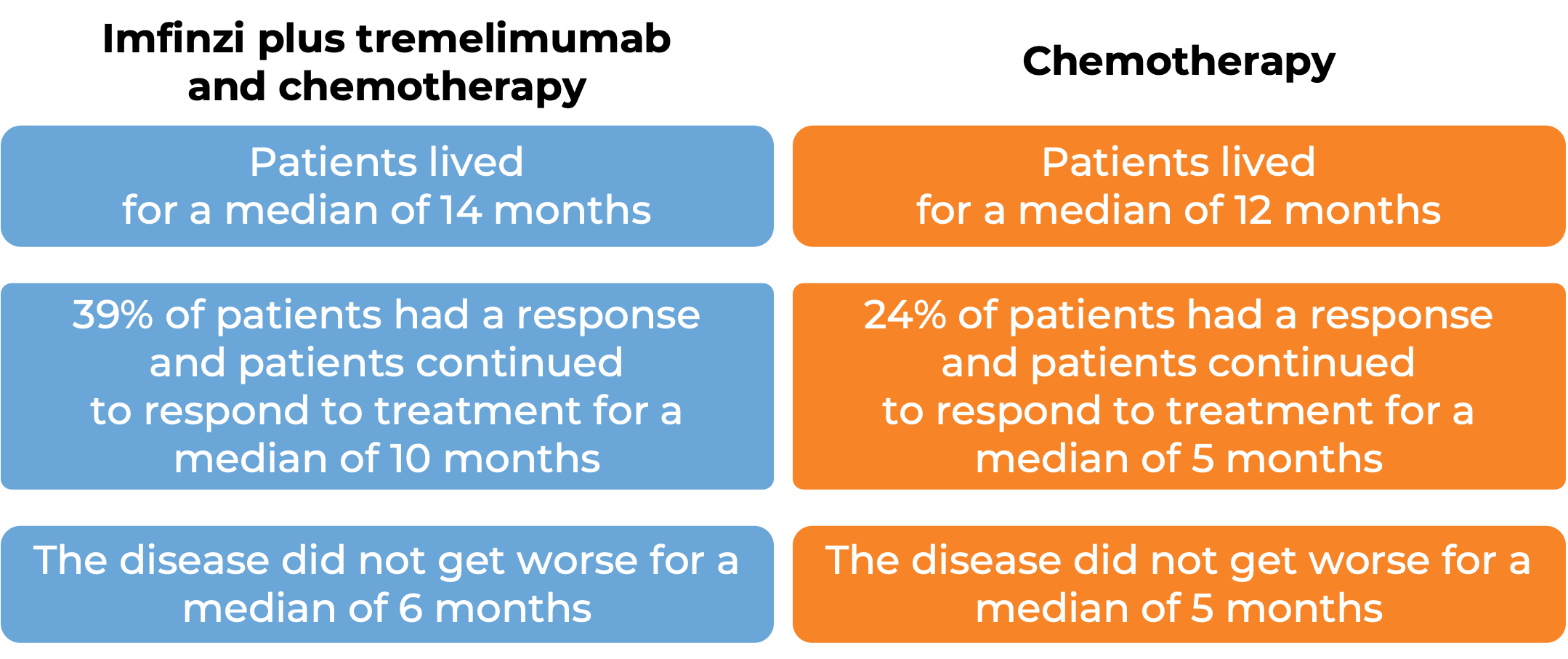
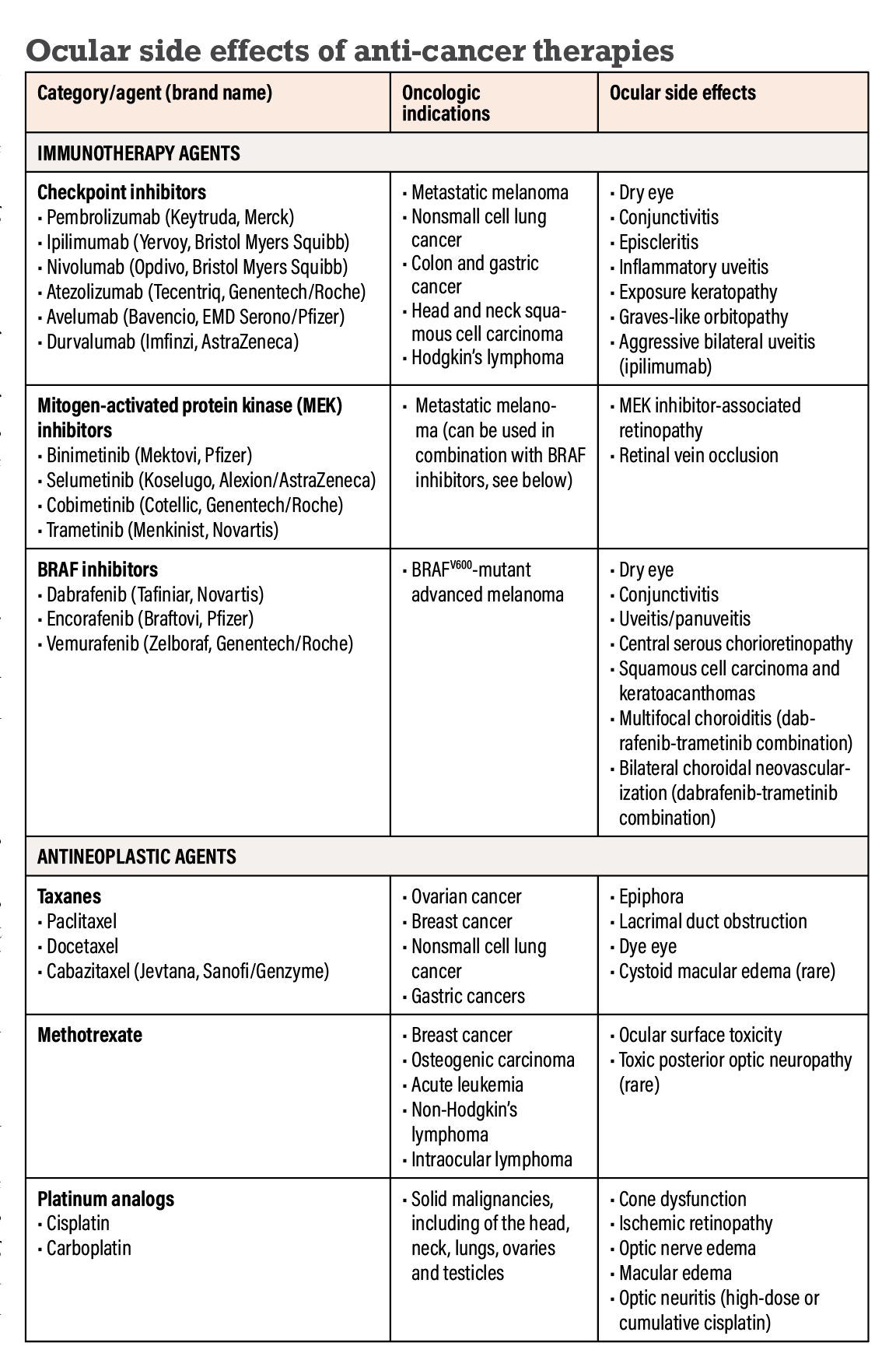
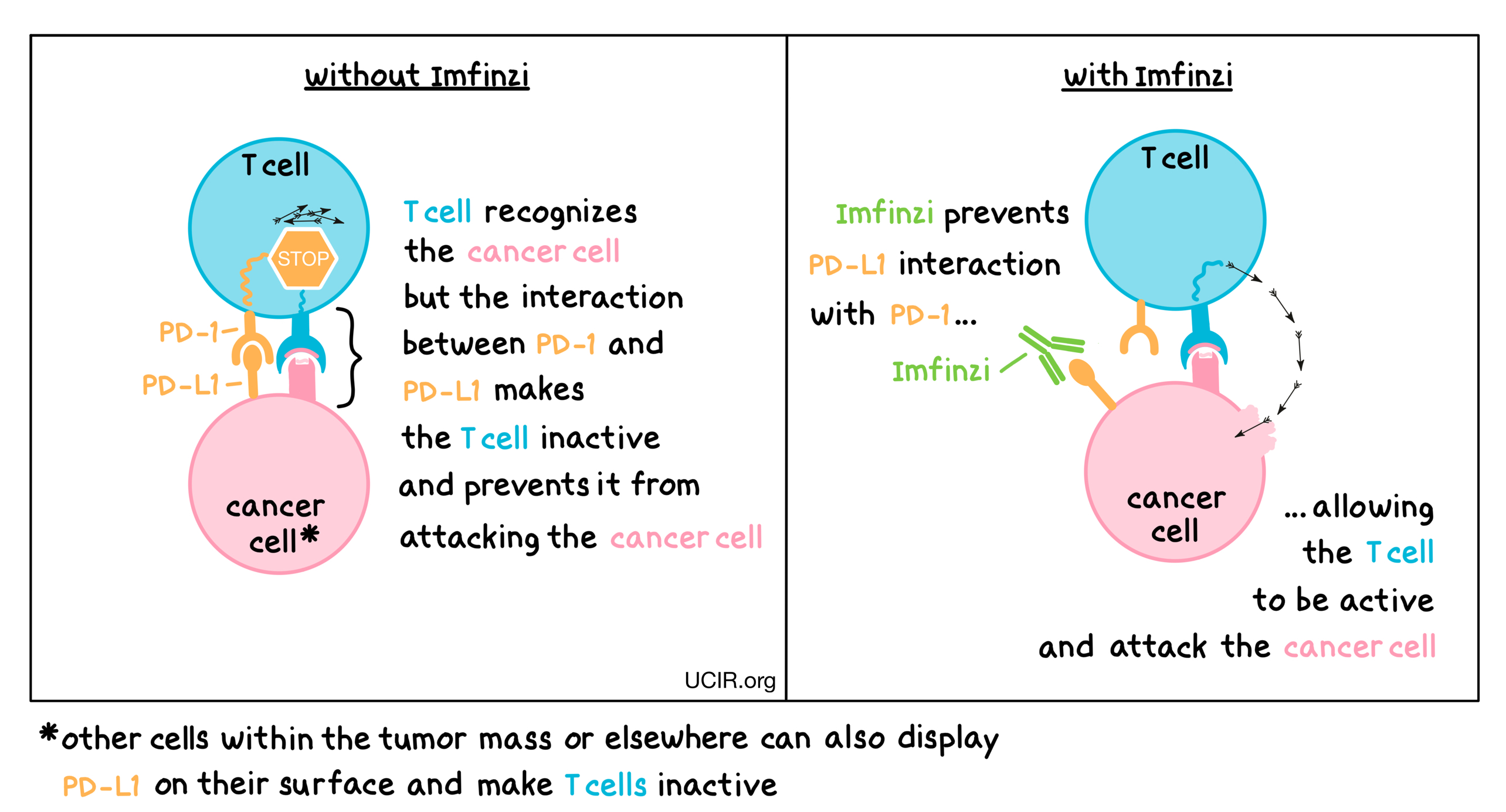
Reimagining Treatment for Unresectable Stage III Non-Small Cell Lung Cancer - Cancer Therapy Advisor

GeparNUEVO: durvalumab plus neoadjuvant chemotherapy in TNBC demonstrates long-term clinical benefit - Onco Americas

Patient-reported outcomes with durvalumab by PD-L1 expression and prior chemoradiotherapy-related variables in unresectable stage III non-small-cell lung cancer | Future Oncology

Durvalumab, with or without tremelimumab, plus platinum–etoposide versus platinum–etoposide alone in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): updated results from a randomised, controlled, open-label, phase 3 trial ...
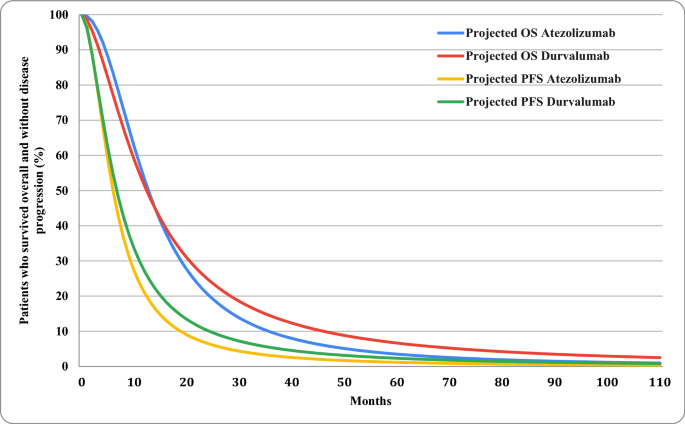
Cost-Effectiveness Analysis of Atezolizumab Versus Durvalumab as First-Line Treatment of Extensive-Stage Small-Cell Lung Cancer in the USA | Clinical Drug Investigation

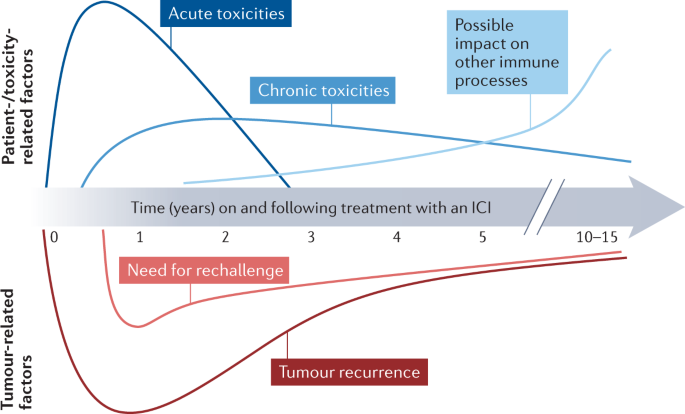
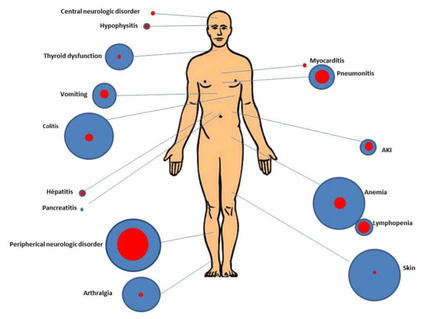
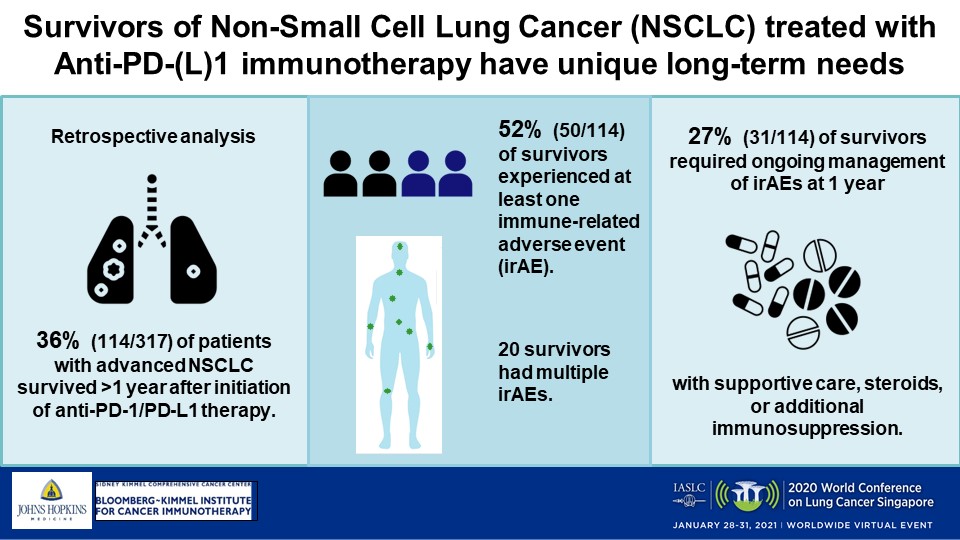
Durvalumab Immunotherapy: Nursing Management of Immune-Related Adverse Events During the Journey of Patients With Stage III Non-Small Cell Lung Cancer | ONS