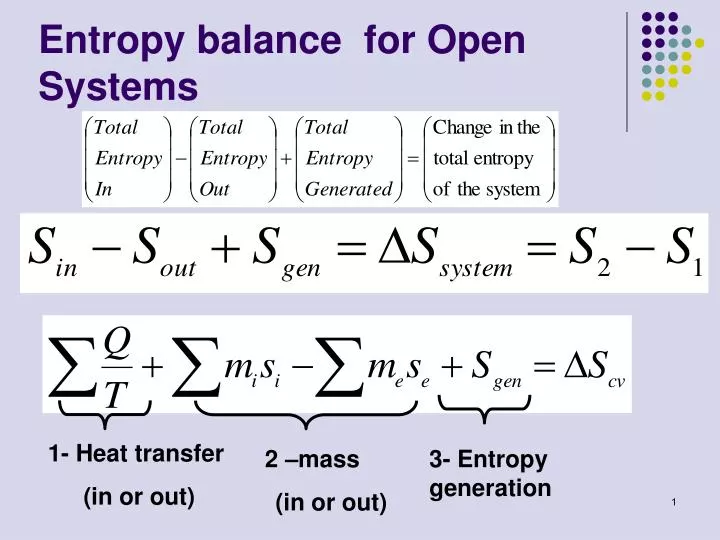
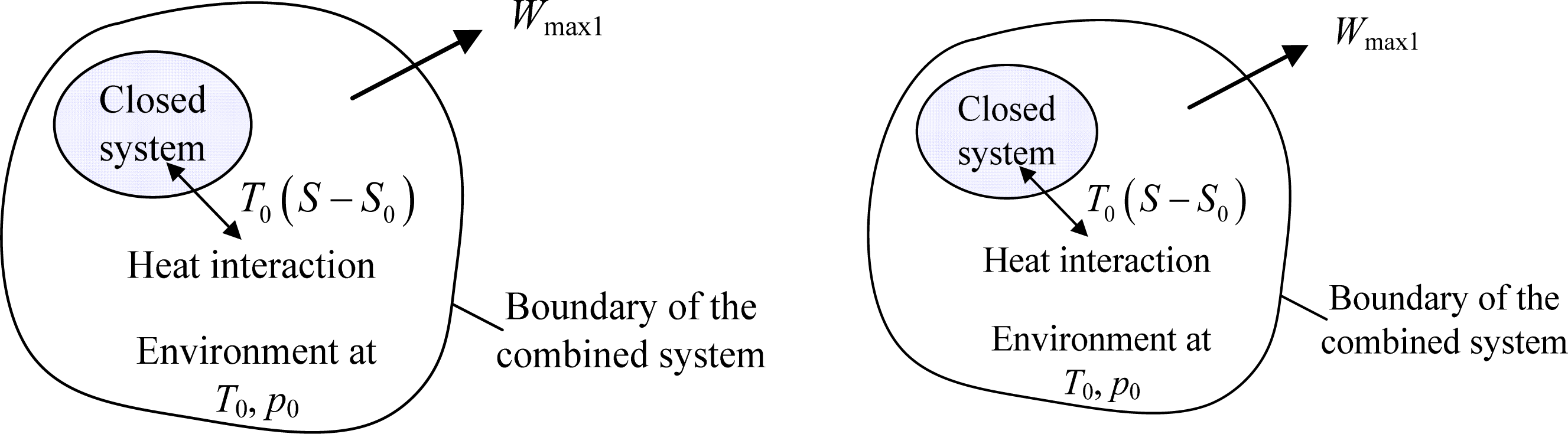
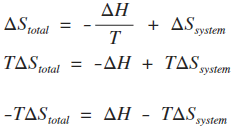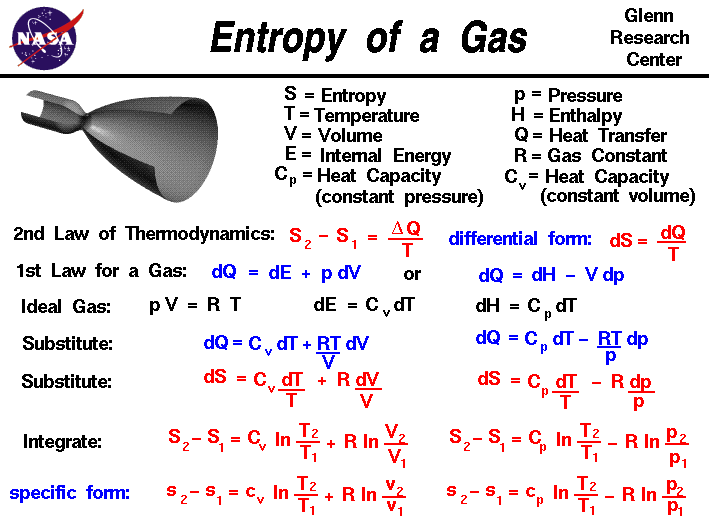SOLVED: The most general formula for the entropy of a system is: S = -kB ln(Ps) In this formula, Ps represents the probability that the system is in a certain state. Assuming

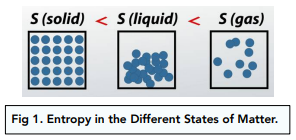
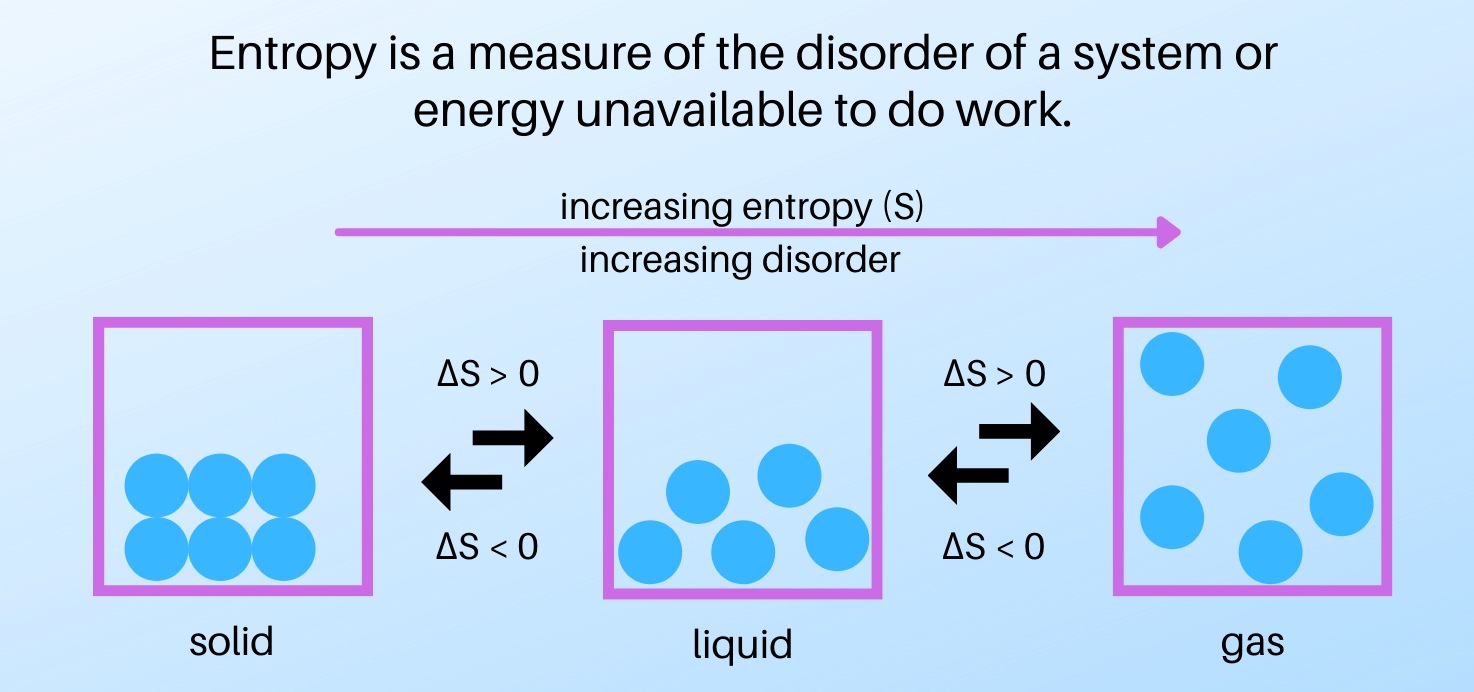
What is Entropy ' A measurement of the degree of randomness of energy in a system. 'The lower the entropy the more ordered and less Examples: gallon of gas, pre… | Teaching

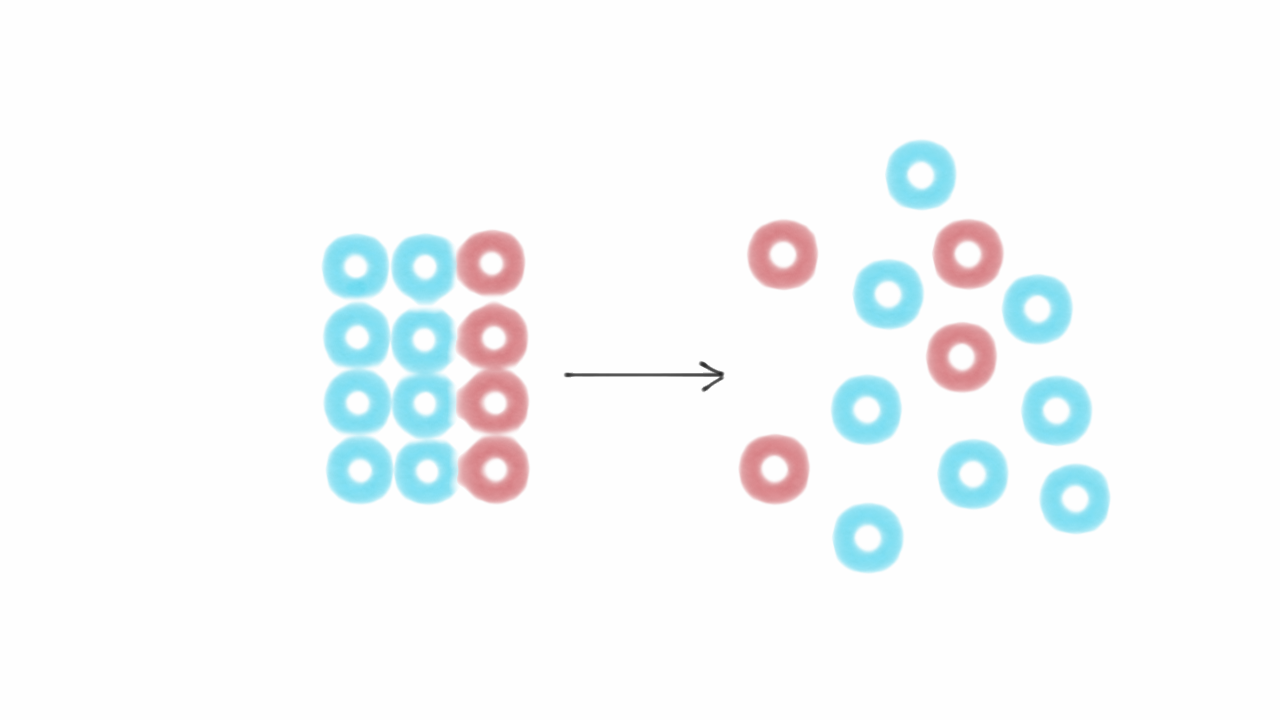
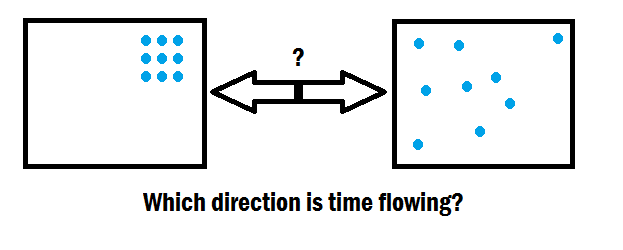
As the entropy of a system increases, it becomes less ordered/increases... | Download Scientific Diagram