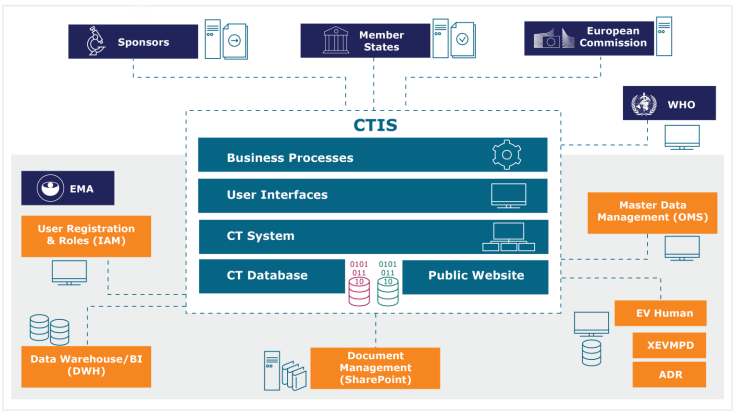
ANSM on X: "31 janv. 2022 entrée en vigueur du règlement européen sur les essais cliniques de médicaments La plateforme Clinical trial information system centralisera le processus de soumission des demandes d'#EssaisCliniques
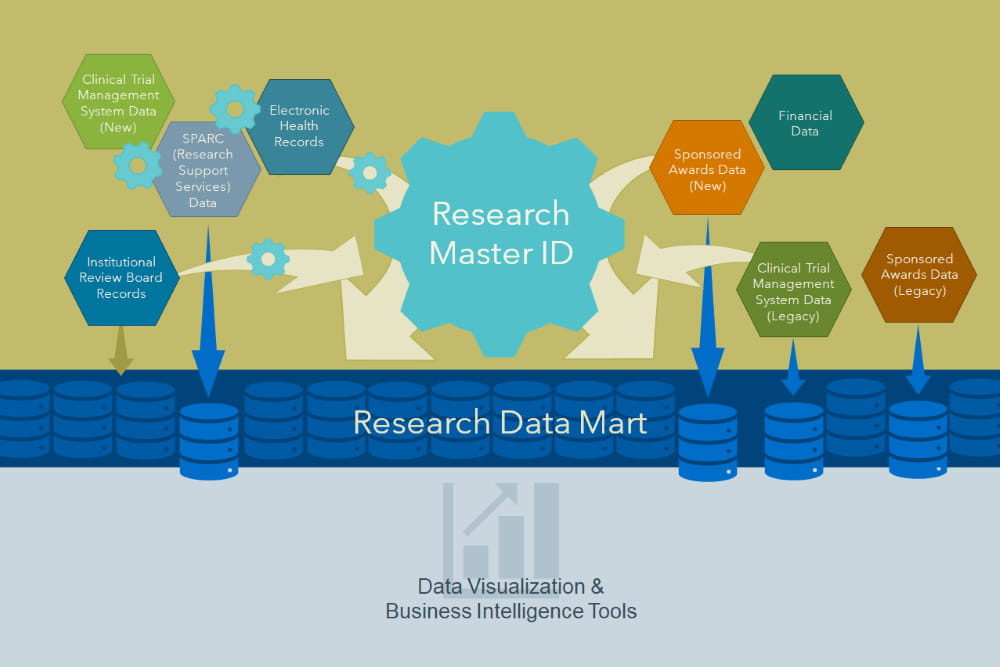
Inspired by data warehousing: A new platform integrates disparate information systems | MUSC | Charleston, SC

In the current system of clinical trials results dissemination, data... | Download Scientific Diagram

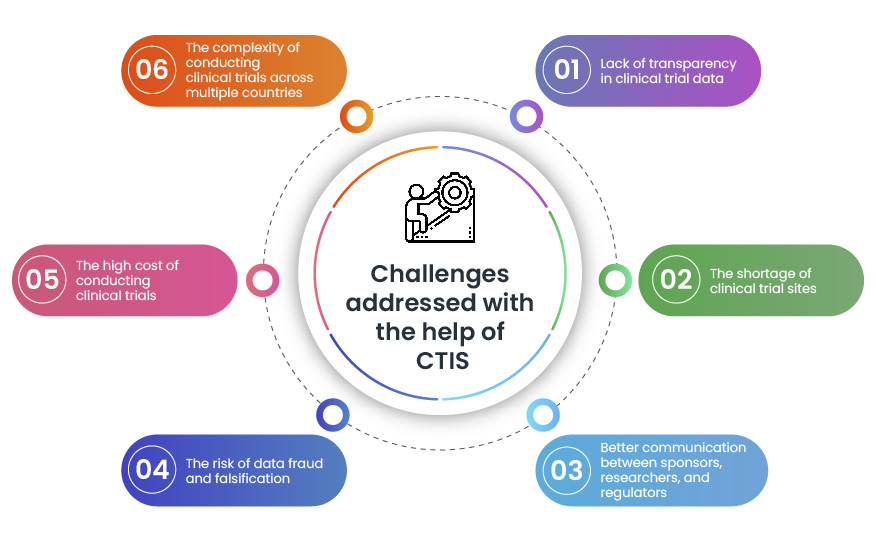
ANSM Agence nationale de sécurité du médicament et des produits de santé sur LinkedIn : Use of Clinical Trials Information System becomes mandatory for new…

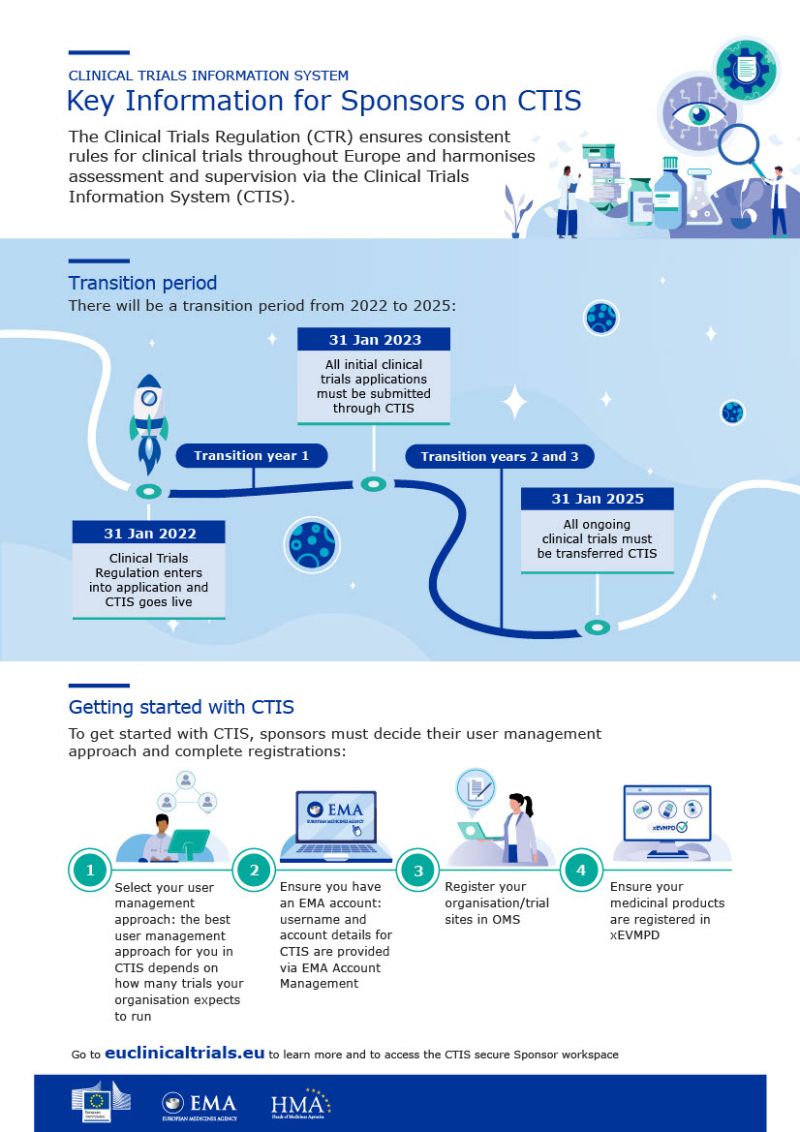
Webinaire EMA le 20/01/2023 : Utilisation du portail CTIS (Clinical Trial Information System) • GIRCI Île-De-France
.jpg)